Implications & Activism
effects of acid rain And what we can do to help
Finally! With a better understanding of chemistry, we can have a more comprehensive discussion about acid rain in the Adirondack Park. We spent the previous section going through the nuts and bolts of acid deposition, and specifically how acidic chemicals are formed and circulated.
However, we have not yet discussed the effects of acid rain, especially in the context of the Adirondack Park. To get the conversation started, here is a list of potential consequences.
-
acidification of lakes and streams
-
damage of trees (especially at high elevations)
-
damage of sensitive forest soils
-
accelerated decay of building materials and paints
-
degradation of visibility (smog)
-
public health implications
Here we will discuss some of the main concerns in more detail. However, the EPA gives some excellent information on these concerns. If you are interested in learning more about topics we do not cover, check out their website: http://www.epa.gov/acidrain/effects/
Acidification of lakes and streams
The effects of acid rain on Adirondack waterways are widespread. Since acids are highly soluble, acidic pollutants are easily dissolved in bodies of water, such as lakes and streams. These pollutants can then be transported via waterways. Additionally, pollutants can be transported in the air.
As mentioned in the "Chemistry Lessons" section, gaseous pollutants (e.g. nitrates, sulfates, and their oxides) are easily mixed with naturally occurring atmospheric gases, and can be transported via wind. These gases then undergo further reactions to form acidic compounds, which can then be deposited in different locations via precipitation. The Adirondack Park is especially at risk for acid rain as shown by wind patterns that show increased acid deposition in the northeast. One of the most acidic ponds reported is Little Echo Pond in Franklin, New York, with a pH of 4.2. Since pH is a logarithmic scale, this means that Little Echo Pond is approximately 1000 times more acidic than a neutral pond (where the pH would be ~7).
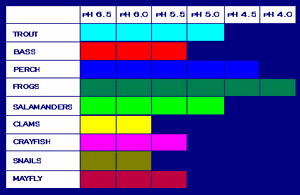
The threat of acid rain to Adirondack waterways, then, is fairly obvious. Acids have an astonishing ability to interact with water and change chemical environments. Acid rain therefore poses a high risk to aquatic species. The chart below, from the EPA’s page, shows the relative sensitivities of different aquatic species to pH.
Fish are only able to maintain their internal pH when in water that has a pH between 6.5 and 8.5. In water with a pH of less than 4, other microorganisms and invertebrates also die off. In the last 100 years, the number of lakes larger than four acres that have a pH less than 5.6 has doubled.
An indirect threat would be an increase in aluminum related to a decrease in pH. As acid rain moves through soils, aluminum is released. Both aluminum and low pH are harmful to fish. Additionally, while some fish are not killed outright by the increased environmental stress, the environmental stress leads to lower body weight, smaller size, and a decreased ability to compete for food and habitat. This example helps to illustrate the cascade of detrimental effects caused by one environmental abnormality.
Effects on forests
Researchers now know that acid rain causes the slowed growth and death of forests. Although forests have some natural defenses against acid deposition (e.g. bicarbonate buffering), soils in the Adirondack region are relatively thin and lacking in minerals. Adirondack soil therefore has a limited ability to neutralize rainfall, especially if the rainfall is more acidic than normal. The effects of this are widespread, and injure trees and other plants.
It is worth mentioning that acid rain does not usually kill trees outright. Instead, acidic environments weaken the trees as a result of limited nutrients, leaf damage, and exposure to toxic substances that released from the soil (e.g. aluminum). Ultimately, trees die as a cumulative result of several threats. Additionally, trees suffer from acid rain even if the soil is well buffered, especially at high elevations. Those trees are often surrounded by acidic clouds and fog that is actually more acidic than rainfall. Essential nutrients in their foliage is stripped away, and they become more susceptible to damage by other environmental factors such as cold winter weather.
Other plants struggle in acidic environments for the same reasons. However, farmed crops usually do not suffer as much as wild plants, because farmers add fertilizer to replace nutrients that the acid would have washed away.
These consequences could be catastrophic to the Adirondack Park and other wilderness areas. This begs the following question:
What can be done?
There is some consolation in that action has already been taken to curb acid deposition. The EPA’s Acid Rain Program (Title IV of the 1970 Clean Air Act) is a comprehensive plan to reduce the effects of acid rain. The plan mandates a two-phase tightening of the restrictions placed on fossil fuel-fired power plants. The plan has been successful in many ways, however the program mainly focuses on sulfur emissions. Sulfur emissions have steadily declined since 1970, but nitrogen concentrations (involved in production of nitric acid) have remained the same, if not increased slightly.
We still have a lot of work to do. As individuals, the best ways to help include the following:
-
driving less frequently (carpooling when possible)
-
conserving energy (turn off lights, do full loads of laundry, take shorter showers, etc.)
-
ACTIVISM
Acid precipitation is a global threat, and unfortunately indviduals can do little to stop it directly. However, activism remains a powerful political tool, and the best way to ignite activism is through education.
In order to make change, it is first important to teach friends and family about the causes and effects of acid precipitation. Don't be afraid to stand up for what you believe in! Share this page, make a Facebook status, tweet about it, or start an eco-activist club at your school. The most important goal: start conversation. Additionally, making contact with local government is an excellent way of reminding legislators that you care, and that environmental issues are still worth addressing.
While we rethink our industrial mindset, we should also adjust our views about life in general, and take responsiblity for the ecological impact we all have. Collectively, we are a nation that depends on electricity. We rely on a variety of electronic devices. We drive everywhere. We forget to turn off the lights. It is important to remember that everyone has an impact on the environment. Our actions start a cascade of environmental effects that can be positive or negative. Positive change has to start somewhere, and hopefully it will start with people like you.